Subscribe to RSS
Please copy the URL and add it into your RSS Feed Reader.
https://www.thieme-connect.de/rss/thieme/en/10.1055-s-00000083.xml
Synlett 2018; 29(02): 181-184
DOI: 10.1055/s-0036-1588578
DOI: 10.1055/s-0036-1588578
letter
Diethylzinc-Mediated Metalloamination–Alkylation of N,N-Dimethylhydrazinoalkenes. Catalysis of C–Zn Alkylation Using Simple Cu(I) Salts
Authors
Generous financial support from the NIGMS (GM116949) is gratefully acknowledged.
Further Information
Publication History
Received: 29 June 2017
Accepted after revision: 01 September 2017
Publication Date:
12 October 2017 (online)
This manuscript is dedicated to Professor Albert Padwa on the occasion of his 80th birthday
Abstract
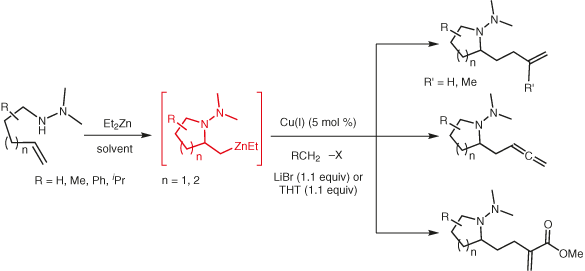
Metalloamination–alkylation of representative N,N-dimethylhydrazinoalkenes has been shown to be effectively catalyzed by CuBr·SMe2, CuCN, and CuI. The current method obviates the use of stoichiometric CuCN(LiCl)2 as a promoter for the electrophilic functionalization event.
Supporting Information
- Supporting information for this article is available online at https://doi.org/10.1055/s-0036-1588578.
- Supporting Information (PDF) (opens in new window)
-
References and Notes
- 1a Meanwell NA. Chem. Res. Toxicol. 2011; 24: 1420
- 1b Welsch ME. Snyder SA. Stockwell BR. Curr. Opin. Chem. Biol. 2010; 14: 347
- 1c Roughley SD. Jordan AM. J. Med. Chem. 2011; 54: 3451
- 2a Sunsdahl B. Smith AR. Livinghouse T. Angew. Chem. Int. Ed. 2014; 53: 14352
- 2b Sunsdahl B. Mickelsen K. Zabawa S. Anderson BK. Livinghouse T. J. Org. Chem. 2016; 81: 10160
- 3a Race NJ. Faulkner A. Fumagalli G. Yamauchi T. Scott JS. Ryden-Landergren M. Sparkes HA. Bower JF. Chem. Sci. 2017; 8: 1981
- 3b Faulkner A. Scott JS. Bower JF. J. Am. Chem. Soc. 2015; 137: 7224
- 3c Turnpenny BW. Chemler SR. Chem. Sci. 2014; 5: 1786
- 3d Liwosz TW. Chemler SR. J. Am. Chem. Soc. 2012; 134: 2020
- 3e Musacchio AJ. Nguyen LQ. Beard GH. Knowles RR. J. Am. Chem. Soc. 2014; 136: 12217
- 3f Choi GJ. Zhu Q. Miller DC. Gu CJ. Knowles RR. Nature 2016; 539: 268
- 3g Choi GJ. Knowles RR. J. Am. Chem. Soc. 2015; 137: 9226
- 3h Garlets ZJ. Parenti KR. Wolfe JP. Chem. Eur. J. 2016; 22: 5919
- 3i White DR. Wolfe JP. J. Am. Chem. Soc. 2015; 137: 11246
- 3j Babij NR. Wolfe JP. Angew. Chem. Int. Ed. 2013; 52: 9247
- 3k Mai DN. Wolfe JP. J. Am. Chem. Soc. 2010; 132: 12157
- 4a Piou T. Rovis T. Nature 2015; 527: 86
- 4b Rong Zhu R. Buchwald SL. J. Am. Chem. Soc. 2015; 137: 8069
- 4c Nicolai S. Sedigh-Zadeh R. Waser J. J. Org. Chem. 2013; 78: 3783
- 4d Nicolai S. Piemontesi C. Waser J. Angew. Chem. Int. Ed. 2011; 50: 4680
- 4e Nicolai S. Waser J. Org. Lett. 2011; 13: 6324
- 4f Hoover JM. DiPasquale A. Mayer JM. Michael FM. J. Am. Chem. Soc. 2010; 132: 5043
- 4g Hoover JM. Freudenthal J. Michael FE. Mayer JM. Organometallics 2008; 27: 2238
- 4h Hewitt JF. M. Williams L. Aggarwal P. Smith CD. France DJ. Chem. Sci. 2013; 4: 3538
- 4i Cernak TA. Lambert TH. J. Am. Chem. Soc. 2009; 131: 3124
- 4j Jana R. Pathak TP. Jensen KH. Sigman MS. Org Lett. 2012; 14: 4074
- 4k Zhang G. Cui L. Wang Y. Zhang L. J. Am. Chem. Soc. 2010; 132: 1474
- 4l Roveda J. Clavette C. Hunt AD. Gorelsky SI. Whipp CJ. Beauchemin AM. J. Am. Chem. Soc. 2009; 131: 8740
- 5 Knochel P. Yeh MC. P. Berk SC. Talbert J. J. Org. Chem. 1988; 53: 2390
- 6 Ogawa A. Curran DP. J. Org. Chem. 1997; 62: 450
- 7 Hoye TR. Eklov BM. Ryba TD. Voloshin M. Yao L. Org. Lett. 2004; 6: 953
- 8 Trost BM. Lautens M. J. Am. Chem. Soc. 1982; 104: 5543
- 9 General Procedure for CuBr·SMe2-Catalyzed Metalloamination–Allylation An oven-dried J. Young tube under an argon atmosphere was charged with Et2Zn (50 μL, 2.0 M in BTF, 0.1 mmol, 1.0 equiv), BTF (0.4 mL), and the requisite N,N-dimethylhydrazinoalkene (0.1 mmol, 1.0 equiv). The tube was then placed into an oil bath (of indicated temperature) and metalloamination–cyclization was monitored using BTF as an internal standard (as evidenced by the disappearance of the alkene signals with concurrent emergence of new C–Zn methylene peaks). The temperature of the reactant mixture was subsequently returned to 23 °C for the addition of THT (0.1 mL) followed by the addition of the Cu(I) catalyst (5 mol%). Allyl bromide (4.0 equiv) was then added and its consumption, with simultaneous recession of the intermediate C–Zn methylene resonance, was monitored by No-D proton NMR. Upon completion of allylation, the reactant mixture was dispersed in Et2O (2 mL) and washed with 1:1 sat. NH4Cl (aq)/sat. aq NH4OH (3 × 3mL). The aqueous layers were then extracted with Et2O (3 mL), and the combined organic layers were dried successively with brine (5 mL) and MgSO4, and then filtered through a plug of silica. To the ethereal solution was added TFA (8.5 μL, 0.11 mmol, 1.1 equiv), and the mixture was concentrated in vacuo. The resulting salt was triturated with pentane (3 × 1 mL), and the volatiles were removed to provide the cyclization–allylation product (>95%) as a clear viscous oil (which could also be isolated as the mono-TFA salt).
For additional references, see:

