Subscribe to RSS
Please copy the URL and add it into your RSS Feed Reader.
https://www.thieme-connect.de/rss/thieme/en/10.1055-s-00000083.xml
Synlett 2013; 24(7): 851-854
DOI: 10.1055/s-0032-1318391
DOI: 10.1055/s-0032-1318391
letter
A Simple and Efficient Synthesis of Dibenzothiophene via BF3·OEt2-Promoted Intramolecular Annulation
Further Information
Publication History
Received: 30 January 2013
Accepted: 11 February 2013
Publication Date:
14 March 2013 (online)
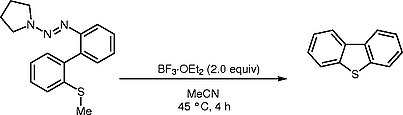

Abstract
A simple and efficient protocol promoted by BF3·OEt2 for synthesizing functionalized dibenzothiophenes (DBTs) has been demonstrated. The annulation condition can tolerate a variety of functional groups.
Supporting Information
- for this article is available online at http://www.thieme-connect.com/ejournals/toc/synlett.
- Supporting Information
-
References and Notes
- 1 Ho TC. Catal. Today 2004; 98: 3
- 2 Gao J, Li L, Meng Q, Li R, Jiang H, Li H, Hu W. J. Mater. Chem. 2007; 17: 1421
- 3 Martin-Santamaria S, Rodriguez J.-J, de Pascual-Teresa S, Gordon S, Bengtsson M, Garrido-Laguna I, Rubio-Viqueira B, Lopez-Casas PP, Hidalgo M, de Pascual-Teresa B, Ramos A. Org. Biomol. Chem. 2008; 6: 3486
- 4a Andrews MD. Sci. Synth. 2000; 10: 211
- 4b Rayner CM, Graham MA. Sci. Synth. 2000; 10: 155
- 4c Gilchrist TL, Higgins SJ. Sci. Synth. 2000; 10: 185
- 4d Gingras M, Raimundo J.-C, Chabre IM. Angew. Chem. Int. Ed. 2006; 45: 1686
- 5 Gilman H, Jacoby AL. J. Org. Chem. 1938; 3: 108
- 6 Sanz R, Fernandez Y, Castroviejo MP, Perez A, Fananas FJ. J. Org. Chem. 2006; 71: 6291
- 7 Kienle M, Unsinn A, Knochel P. Angew. Chem. Int. Ed. 2010; 49: 4751
- 8 Takashi O, Mayumi F, Daiki S, Keiji M. Adv. Synth. Catal. 2001; 343: 166
- 9 Samanta R, Antonchick AP. Angew. Chem. Int. Ed. 2011; 50: 5217
- 10 Jepsen TH, Larsen M, Jorgensen M, Solanko KA, Bond AD, Kadziola A, Nielsen MB. Eur. J. Org. Chem. 2011; 1: 53
- 11 Korang J, Grither WR, McCulla RD. J. Am. Chem. Soc. 2010; 132: 4466
- 12 Nayak PK, Agarwal N, Periasamy N. J. Chem. Sci. 2010; 122: 119
- 13 Kimball DB, Haley MM. Angew. Chem. Int. Ed. 2002; 41: 3338
- 14 Liu C, Knochel P. J. Org. Chem. 2007; 72: 7106
- 15a Yang W, Zhou J, Wang B, Ren H. Chem. Eur. J. 2011; 17: 13665
- 15b Zhao G, Wang B, Yang W, Ren H. Eur. J. Org. Chem. 2012; 31: 6236
- 16 Zhou J, Yang W, Wang B, Ren H. Angew. Chem. Int. Ed. 2012; 51: 12293
- 17a Haraguchi H, Ishikawa H, Mizutani K, Tamura Y, Kinoshita T. Bioorg. Med. Chem. 1998; 6: 339
- 17b Vogel S, Heilmann J. J. Nat. Prod. 2008; 71: 1237
- 17c Edwards ML, Stemerick DM, Sunkara PS. J. Med. Chem. 1990; 33: 1948
- 17d Ducki S, Rennison D, Woo M, Kendall A, Chabert JF. D, Mcgown AT, Lawrence NJ. Bioorg. Med. Chem. 2009; 17: 7698
- 18a Beadle JR, Korzeniowski SH, Rosenberg DE, Garcia-Slanga BJ, Gokel GW. J. Org. Chem. 1984; 49: 1594
- 18b Patrick TB, Willaredt RP, DeGonia DJ. J. Org. Chem. 1985; 50: 2232
- 18c Satyamurthy N, Barria JR, Schmidt DG, Kammerer C, Bida GT, Phelps ME. J. Org. Chem. 1990; 55: 4560
- 18d Saeki T, Son E.C, Tamao K. Bull. Chem. Soc. Jpn. 2005; 78: 1654
- 19 Pandya VB, Jain MR, Chaugule BV, Patel JS, Parmar BM, Joshi JK, Patel PR. Synth. Commun. 2012; 42: 497
- 20 Representative Procedure for the Synthesis of 3a: BF3·OEt2 (2.0 equiv) was added dropwise to the stirred solution of 2a (1 mmol) in anhyd MeCN at 45 °C. The mixture was stirred for 4 h. After the completion of the reaction detected by TLC, it was washed with sat. aq NaHCO3 and extracted with EtOAc. The combined organic extracts were dried over anhyd Na2SO4 and removed under reduced pressure. The product was purified by column chromatography to give 3a as a white solid (80% yield); mp 68–71 °C. 1H NMR (400 MHz, CDCl3): δ = 8.84 (s, 1 H), 8.22–8.26 (m, 1 H), 8.11–8.14 (m, 1 H), 7.84–7.90 (m, 2 H), 7.48–7.51 (m, 2 H), 4.46 (q, J = 6.8 Hz, 2 H), 1.47 (t, J = 7.2 Hz, 3 H). 13C NMR (100 MHz, CDCl3): δ = 166.7, 144.2, 139.6, 135.5, 135.2, 127.3, 126.8, 124.8, 123.1, 122.8, 122.5, 121.9, 61.1, 14.4. IR (thin film): 3065, 2924, 1710, 1267, 1100, 1025 cm–1. MS (EI): m/z (%) = 256 (97) [M+], 241 (14), 228 (39), 211 (100), 183 (58), 139 (54). HRMS (EI–TOF): m/z [M+] calcd for C15H12O2S: 256.0558; found: 256.0566.
