Subscribe to RSS
DOI: 10.1055/s-0037-1610346
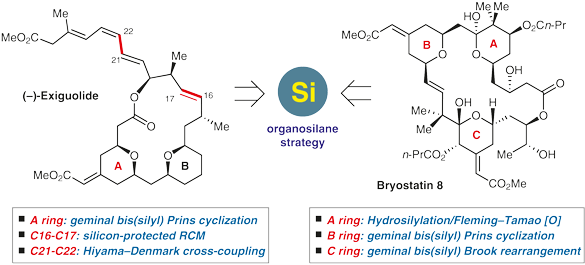
Total Synthesis of Bryostatin 8 and (–)-Exiguolide: Applications of an Organosilane Strategy
We are grateful for financial support from the NSFC (21622202, 21290180) and the MOST (2017ZX09101003-005-004).Publication History
Received: 08 November 2018
Accepted after revision: 08 November 2018
Publication Date:
18 December 2018 (online)

Dedication to Professor Yong-Qiang Tu on the occasion of his 60th birthday
Abstract
In this Account, we give an overview of our recent efforts towards the total synthesis of two marine natural products: bryostatin 8 and (–)-exiguolide. The central theme is to highlight the power of the organosilane strategy, which was characterized with our geminal bis(silanes)-based methodologies for constructing the unique exocyclic enoate-substituted pyran rings.
1 Introduction
2 Background of Bryostatins and (–)-Exiguolide
3 Constructing the B-Ring of Bryostatins
4 Total Synthesis of (–)-Exiguolide via an Organosilane Strategy
5 Constructing the C-Ring of Bryostatins
6 Total Synthesis of Bryostatin 8
7 Conclusion
-
References and Notes
- 1a Overman LE, Blumenkopf TA. Chem. Rev. 1986; 86: 857
- 1b Panek M, Masse CE. Chem. Rev. 1995; 95: 1293
- 1c Langkopf E, Schinzer D. Chem. Rev. 1995; 95: 1375
- 1d Mikael B, Troels S. Chem. Rev. 1995; 95: 1253
- 1e Fleming I, Barbero A, Walter D. Chem. Rev. 1997; 97: 2063
- 1f Mi Y, Schreiber JV, Corey EJ. J. Am. Chem. Soc. 2002; 124: 11290
- 1g Linghu X, Johnson JS. Angew. Chem. Int. Ed. 2003; 42: 2534
- 1h Friese JC, Krause S, Schafer HJ. Tetrahedron Lett. 2002; 43: 2683
- 1i Sun P, Sun C, Weinreb SM. J. Org. Chem. 2002; 67: 4337
- 1j Denmark SE, Yang SM. J. Am. Chem. Soc. 2002; 124: 15196
- 1k Rychnovsky SD, Khire UR, Yang G. J. Am. Chem. Soc. 1997; 119: 2058
- 1l Toro A, Nowak P, Deslongchamps P. J. Am. Chem. Soc. 2000; 122: 4526
- 1m Wender PA, Hegde SG, Hubbard RD, Zhang L. J. Am. Chem. Soc. 2002; 124: 4956
- 1n Hale KJ, Hummersone MG, Bhatia GS. Org. Lett. 2000; 2: 2189
- 1o Chen CL, Sparks SM, Martin SF. J. Am. Chem. Soc. 2006; 128: 13696
- 2a Lautens M, Delanghe PH. M, Goh JB, Zhang CH. J. Org. Chem. 1992; 57: 3270
- 2b Lautens M, Delanghe PH. M, Goh JB, Zhang CH. J. Org. Chem. 1995; 60: 4213
- 2c Williams DR, Morales-Ramos ÁI, Williams CM. Org. Lett. 2006; 8: 4393
- 2d Luo Q, Wang C, Li YX, Ouyang KB, Gu L, Uchiyama M, Xi ZF. Chem. Sci. 2011; 2: 2271
- 2e Li H, Liu LT, Wang ZT, Zhao F, Zhang SG, Zhang WX, Xi ZF. Chem. Eur. J. 2011; 17: 7399
- 2f Groll K, Manolikakes SM, du Jourdin XM, Jaric M, Bredihhin A, Karaghiosoff K, Carell T, Knochel P. Angew. Chem. Int. Ed. 2013; 52: 6776
- 2g Cui HY, Zhang JY, Cui CM. Organometallics 2013; 32: 1
- 2h Werner V, Klatt T, Fujii M, Markiewicz J, Apeloig Y, Knochel P. Chem. Eur. J. 2014; 20: 8338
- 2i Liu ZX, Tan HC, Fu TR, Xia Y, Qiu D, Zhang Y, Wang JB. J. Am. Chem. Soc. 2015; 137: 12800
- 2j Das M, Manvar A, Jacolot M, Blangetti M, Jones RC, O'Shea DF. Chem. Eur. J. 2015; 21: 8737
- 2k Das M, O'Shea DF. Org. Lett. 2016; 18: 336
- 2l Maudrich J.-J, Sindlinger CP, Aicher FS. W, Eichele K, Schubert H, Wesemann L. Chem. Eur. J. 2017; 23: 2192
- 2m Mizuhata Y, Fujimori S, Sasamori T, Tokitoh N. Angew. Chem. Int. Ed. 2017; 56: 4588
- 2n Tanabe M, Hagio T, Osakada K, Nakamura M, Hayashi Y, Ohshita J. Organometallics 2017; 36: 1974
- 3a Liu ZJ, Lin XL, Yang N, Su ZS, Hu CW, Xiao PH, He YY, Song ZL. J. Am. Chem. Soc. 2016; 138: 1877
- 3b Chu ZW, Wang K, Gao L, Song ZL. Chem. Commun. 2017; 53: 3078
- 3c Chu Y, Pu Q, Tang ZX, Gao L, Song ZL. Tetrahedron 2017; 73: 3707
- 3d Pu Q, Tang XX, Gao L, Song ZL. Org. Chem. Front. 2018; 5: 2035
- 4a Li HZ, Xie HM, Zhang ZG, Xu YJ, Lu J, Gao L, Song ZL. Chem. Commun. 2015; 40: 8484
- 4b Zhang YB, Guo QY, Sun XW, Lu J, Cao YJ, Pu Q, Chu ZW, Gao L, Song ZL. Angew. Chem. Int. Ed. 2018; 57: 942
- 5a Hale KJ, Hummersone MG, Manaviazar S, Frigerio M. Nat. Prod. Rep. 2002; 19: 413
- 5b Kortmansky J, Schwartz GK. Cancer Invest. 2003; 21: 924
- 5c Wender PA, Baryza JL, Hilinski MK, Horan JC, Kan C, Verma VA. Beyond Natural Products: Synthetic Analogues of Bryostatin 1 . In Drug Discovery Research: New Frontiers in the Post-Genomic Era . Huang Z. John Wiley and Sons; Hoboken, NJ: 2007: 127
- 5d Hale KJ, Manaviazar S. Chem. Asian J. 2010; 5: 704
- 5e Lu Y, Krische MJ. Bryostatin 7 . In Total Synthesis of Naural Products . Li J, Corey EJ. Springer; Berlin, Heidelberg: 2012: 103
- 5f Wender PA. Rethinking the Role of Natural Products: Function-Oriented Synthesis, Bryostatin, and Bryologs. In Products in Medicinal Chemistry. Hanessian S. Wiley-VCH; Weinheim: 2014: 475
- 5g Kollár P, Rajchard J, Balounová Z, Pazourek J. Pharm. Biol. 2014; 52: 237
- 5h Tian XR, Tang HF, Tian XL, Hu JJ, Huang LL, Gustafson KR. Future Med. Chem. 2018; 10: 1497
- 6 Pettit GR, Herald CL, Doubek DL, Herald DL, Arnold E, Clardy J. J. Am. Chem. Soc. 1982; 104: 6846
- 7 Yu HB, Yang F, Sun F, Ma GY, Gan JH, Hu WZ, Han BN, Jiao WH, Lin HW. J. Nat. Prod. 2015; 78: 1169
- 8 Kortmansky J, Schwartz GK. Cancer Invest. 2003; 21: 924
- 9 Schwartz GK, Shah MA. J. Clin. Oncol. 2005; 23: 9408
- 10 Sun MK, Hongpaisan J, Nelson TJ, Alkon DL. Proc. Natl. Acad. Sci. U.S.A. 2008; 105: 13620
- 11 Way KJ, Katai N, King GL. Diabet. Med. 2001; 18: 945
- 12a Williams P, Sorribas A, Howes M.-JR. Nat. Prod. Rep. 2011; 28: 48
- 12b Xu C, Liu Q.-Y, Alkon DL. Neuroscience 2014; 268: 75
- 12c Nelson TJ, Sun MK, Lim C, Sen A, Khan T, Chirila FV, Alkon DL. J. Alzheimer’s Dis. 2017; 58: 521
- 13a Archin NM, Margolis DM. Curr. Opin. Infect. Dis. 2014; 27: 29
- 13b Laird GM, Bullen CK, Rosenbloom DI. S, Martin AR, Hill AL, Durand CM, Siliciano JD, Siliciano RF. J. Clin. Invest. 2015; 125: 1901
- 14 Krische MJ, Ketcham JM, Volchkov I, Chen TY, Blumberg PM, Kedei N, Lewin NE. J. Am. Chem. Soc. 2016; 138: 13415 ; and references therein
- 15a Kazanietz MG, Lewin NE, Gao F, Petit GR, Blumberg PM. Mol. Pharmacol. 1994; 46: 374
- 15b Griner EM, Kazanietz MG. Nat. Rev. Cancer 2007; 7: 281
- 15c Lew DJ, Rout MP. Curr. Opin. Cell Biol. 2009; 21: 1
- 16 Schaufelberger DE, Koleck MP, Beutler JA, Vatakis AM, Alvarado AB, Andrews P, Marzo LV, Muschik GM, Roach J, Ross JT. J. Nat. Prod. 1991; 54: 1265
- 17a Wender PA, Nakagawa Y, Near KE, Staveness D. Org. Lett. 2014; 16: 5136
- 17b Keck GE, Kraft MB, Poudel YB, Kedei N, Lewin NE, Peach ML, Blumberg PM. J. Am. Chem. Soc. 2014; 136: 13202
- 17c Krische MJ, Andrews IP, Ketcham JM, Blumberg PM, Kedei N, Lewin NE, Peach ML. J. Am. Chem. Soc. 2014; 136: 13209
- 17d Keck GE, Kelsey JS, Cataisson C, Chen J, Herrmann MA, Petersen ME, Baumann DO, McGowan KM, Yuspa SH, Blumberg PM. Mol. Carcinog. 2016; 55: 2183
- 17e Marsden MD, Wu XM, Navab SM, Loy BA, Schrier AJ, DeChristopher BA, Shimizu AJ, Hardman CT, Ho S, Ramirez CM, Wender PA, Zack JA. Virology. 2018; 520: 83
- 17f Khan TK, Wender PA, Alkon DL. J. Cell Physiol. 2018; 233: 1523
- 17g Zhao XG, Kedei N, Michalowski A, Lewin NE, Keck GE, Blumberg PM. ChemBioChem. 2018; 19: 1049
- 17h Cummins TJ, Kedei N, Czikora A, Lewin NE, Kirk S, Petersen ME, McGowan KM, Chen JQ, Luo X, Johnson RC, Ravichandran S, Blumberg PM, Keck GE. ChemBioChem 2018; 19: 877
- 18a Kageyama M, Tamura T, Nantz MH, Roberts JC, Somfai P, Whritenour DC, Masamune S. J. Am. Chem. Soc. 1990; 112: 7407
- 18b Evans DA, Carter PH, Carreira EM, Prunet JA, Charette AB, Lautens M. Angew. Chem. Int. Ed. 1998; 37: 2354
- 18c Evans DA, Carter PH, Carreira EM, Prunet JA, Charette AB, Lautens M. J. Am. Chem. Soc. 1999; 121: 7540
- 18d Ohmori K, Ogawa Y, Obitsu T, Ishikawa Y, Nishiyama S, Yamamura S. Angew. Chem. Int. Ed. 2000; 39: 2290
- 18e Aliev AE, Hale KJ. Org. Lett. 2006; 8: 4477
- 18f Trost BM, Dong G. Nature 2008; 456: 485
- 18g Trost BM, Dong G. J. Am. Chem. Soc. 2010; 132: 16403
- 18h Keck GE, Poudel YB, Cummins TJ, Rudra A, Covel JA. J. Am. Chem. Soc. 2011; 133: 744
- 18i Wender PA, Schrier AJ. J. Am. Chem. Soc. 2011; 133: 9228
- 18j Lu Y, Woo SK, Krische MJ. J. Am. Chem. Soc. 2011; 133: 13876
- 18k Wender PA, Hardman CT, Ho S, Jeffreys MS, Maclaren JK, Quiroz RV, Ryckbosch SM, Shimizu AJ, Sloane JL, Stevens MC. Science 2017; 358: 218
- 19a Ohta S, Uy MM, Yanai M, Ohta E, Hirata T, Ikegami S. Tetrahedron Lett. 2006; 47: 1957
- 19b Kwon MS, Woo SK, Na SW, Lee E. Angew. Chem. Int. Ed. 2008; 47: 1733
- 19c Fuwa H, Sasaki M. Org. Lett. 2010; 12: 584
- 19d Cook C, Guinchard X, Liron F, Roulland E. Org. Lett. 2010; 12: 744
- 19e Crane EA, Zabawa TP, Farmer RL, Scheidt KA. Angew. Chem. Int. Ed. 2011; 50: 9112
- 19f Fuwa H, Suzuki T, Kubo H, Yamori T, Sasaki M. Chem. Eur. J. 2011; 17: 2678
- 19g Cook C, Liron F, Guinchard X, Roulland E. J. Org. Chem. 2012; 77: 6728
- 20 Cossy J. C. R. Chim. 2008; 11: 1477
- 21 Tanaka K, Otsubo K, Fuji K. Tetrahedron Lett. 1996; 37: 3735
- 22a Crane EA, Scheidt KA. Angew. Chem. Int. Ed. 2010; 49: 8316
- 22b Han X, Peh GR, Floreancig PE. Eur. J. Org. Chem. 2013; 1193
- 22c Greco SJ, Fiorot RG, Lacerda V, dos Santos RB. Aldrichimica Acta 2013; 46: 59
- 23 Lu J, Song ZL, Zhang YB, Gan ZB, Li HZ. Angew. Chem. Int. Ed. 2012; 51: 5367
- 24a Gradillas A, Pérez-Castells J. Synthesis of Natural Products Containing Macrocycles by Alkene Ring-Closing Metathesis. In Metathesis in Natural Product Synthesis, Strategies, Substrates, and Catalyst. Cossy J, Arseniyadis S, Meyer C. Wiley-VCH; Weinheim: 2010: 149
- 24b Nelson DJ, Manzini S, Urbina-Blanco CA, Nolan SP. Chem. Commun. 2014; 50: 10355
- 24c Hoveyda AH. J. Org. Chem. 2014; 79: 4763
- 24d Gaddam J, Mallurwar NK, Konda S, Khatravath M, Aeluri M, Mitra P, Arya P. Ring-Closing Metathesis-Based Methods in Chemical Biology. In Practical Medicinal Chemistry with Macrocycles . Marsault E, Peterson ML. John Wiley and Sons; Hoboken, NJ: 2017: 243
- 24e Lecourt C, Dhambri S, Allievi L, Sanogo Y, Zeghbib N, Othman RB, Lannou M.-I, Sorin G, Ardisson J. Nat. Prod. Rep. 2018; 35: 105
- 25 Garber SB, Kingsbury JS, Gray BL, Hoveyda AH. J. Am. Chem. Soc. 2000; 122: 8168
- 26a Hiyama T. Organosilicon Compounds in Cross-Coupling Reactions. In Metal-Catalyzed Cross-Coupling Reactions . Diederich F, Stang PJ. Wiley-VCH; Weinheim: 1998. Chap. 1
- 26b Hiyama T, Shirakawa E. Top. Curr. Chem. 2002; 219: 61
- 26c Denmark SE, Regens CS. Acc. Chem. Res. 2008; 41: 1486
- 26d Chang WT. T, Smith RC, Regens CS, Bailey AD, Werner NS, Denmark SE. Cross-Coupling with Organosilicon Compounds. In Organic Reactions . Denmark SE. John Wiley and Sons; Hoboken, NJ: 2011. Chap.
- 26e Denmark SE, Liu H.-C. Angew. Chem. Int. Ed. 2010; 49: 2978
- 26f Nakao Y, Hiyama T. Chem. Soc. Rev. 2011; 40: 4893
- 26g Sore HF, Galloway WR. J. D, Spring DR. Chem. Soc. Rev. 2012; 41: 1845
- 27a Ikegashira K, Nishihara Y, Hirabayashi K, Mori A, Hiyama T. Chem. Commun. 1997; 33: 1039
- 27b Denmark SE, Kobayashi T. J. Org. Chem. 2003; 68: 5153
- 28 Lu J, Zhang YB, Yang WY, Guo QY, Gao L, Song ZL. Org. Lett. 2017; 19: 5232
- 29a Espino CG, Wehn PM, Chow J, Du Bois J. J. Am. Chem. Soc. 2001; 123: 6935
- 29b Espino CG, Du Bois J. Angew. Chem. Int. Ed. 2001; 40: 598
- 29c Fleming JJ, Du Bois J. J. Am. Chem. Soc. 2006; 128: 3926
- 30 Trost BM, Yang H, Thiel OR, Frontier AJ, Brindle CS. J. Am. Chem. Soc. 2007; 129: 2206
- 31 Gao L, Lu J, Song ZL, Lin XL, Xu YJ, Yin ZP. Chem. Commun. 2013; 49: 8961
- 32a Brook AG. Acc. Chem. Res. 1974; 7: 77
- 32b Moser WH. Tetrahedron 2001; 57: 2065
- 32c Kira M, Iwamoto T. Silyl Migrations. In The Chemistry of Organic Silicon Compounds . Rappoport Z, Apeloig Y. John Wiley and Sons; Chichester: 2001: 853
- 32d Smith III AB, Adams CM. Acc. Chem. Res. 2004; 37: 365
- 32e Schaumann E, Kirschning A. Synlett 2007; 177
- 33a Tamao K, Akita M, Kumada M. J. Organomet. Chem. 1983; 254: 13
- 33b Fleming I, Henning R, Plaut H. J. Chem. Soc., Chem. Commun. 1984; 29
- 33c Jones GR, Landais Y. Tetrahedron 1996; 52: 7599
- 34 Evans DA, Hoveyda AH. J. Am. Chem. Soc. 1990; 112: 6447
- 35 Tamao K, Sumitani K, Kumada M. J. Am. Chem. Soc. 1972; 94: 4374
- 36 Okazoe T, Takai K, Utimoto K. J. Am. Chem. Soc. 1987; 109: 951
- 37 Hentges SG, Sharpless KB. J. Am. Chem. Soc. 1980; 102: 4263
- 38 Inanaga J, Hirata K, Saeki H, Katsuki T, Yamaguchi M. Bull. Chem. Soc. Jpn. 1979; 52: 1989
For selective reviews on organosilanes, see:
Brook rearrangement – for selected examples, see:
Danheiser cyclopentene annulation:
Fleming–Tamao oxidation:
Hiyama-Denmark Cross coupling:
Mukaiyama aldol:
Peterson olefination:
Sakurai allylation:
Smith–Tietze multicomponent dithiane linpinch coupling:
Silicon-tethered strategy:
For latest progresses achieved in our group, see:
For reviews on the bryostatins, see:
For the latest studies on bryostatin analogues, see:
For total synthesis of the bryostatins, see for bryostatin 7:
Bryostatin 2:
Bryostatin 3:
Formal synthesis of bryostatin 7:
Bryostatin 16:
Bryostatin 1:
Bryostatin 9:
Bryostatin 7:
Bryostatin 1:
For total synthesis, see:
For selected reviews, see:
For the latest reviews on RCM reactions, see:
For selected reviews, see:
For selected works, see:
For selected reviews on the Brook rearrangement, see:
For a review on Fleming–Tamao oxidation, see:
